By Alexey Aksenov, Alexander Mitilineos, Alexander Polyakov, PhD and Joseph L. Shmidt, PhD

Activated carbon (AC) cartridges are widely used in POU and POE water purification applications for:
• Prefiltration before RO membranes for reducing active chlorine, organics, particulates and colloids (including rust), which may plug up and/or damage RO membranes
• Postfiltration after RO for reducing the amount of small, mostly organic molecules not removed by RO membranes and elimination of unpleasant taste and odors (mostly organic in nature) leaching into water from the material of the RO permeate water tank
 • Direct-flow adsorption filters for removing active chlorine, organics both VOC and larger molecular-weight compounds, suspended solids including rust, heavy metals and other toxic small-sized and/or complex, larger compounds.
• Direct-flow adsorption filters for removing active chlorine, organics both VOC and larger molecular-weight compounds, suspended solids including rust, heavy metals and other toxic small-sized and/or complex, larger compounds.
Activated carbon cartridges are comprised of either granular activated carbon (GAC) or carbon blocks (powdered activated carbon [PAC]) fused with powdered polyethylene into an annular, cylindrical shape. In the carbon blocks (CB), water flows radially from the outside to the inner annular opening. As compared to granular carbon cartridges, carbon blocks have larger dirt-holding capacities and are capable of working at high flowrates. Such carbon blocks may have a larger annular outer surface area, short flow path and can be made of fine AC powder. It is not possible to use this AC powder in the regular axial-flow cartridges due to high flow resistance.
Carbon blocks are made by either compression molding or extruding a mixture of PAC and powdered polyethylene. Inherently adhesive high molecular-weight polyethylene particles soften, start to melt and bind PAC particles into a matrix. Additional capabilities (such as lead removal) are achieved by adding specialized adsorbents (e.g., activated alumina) to the mixture. Because of the relatively high processing temperature, many water purification materials (e.g., ion-exchange resin) cannot be used. Properties of the carbon blocks may be varied by changing the particular size distribution of the PAC and degree of compression during the formation process. (Carbon blocks have somewhat less adsorptive capacity than GAC because the adhesive [PE] occupies some of the sites.)
Study details
Comparative testing was conducted according to NSF/ANSI-42/53 standards protocols. General test water was prefiltered through a 5-micron absolute-rating filter (temperature 20 ± 2°C; pH 7.5 ± 0.5; turbidity < 1 NTU; TDS 300 ± 100 ppm; hardness 50 ppm). Test conditions included flowrate of 1.5 L/m, based upon 50-percent on/50 percent off with a 20-minute cycle, 16 hours per day, followed by an eight-hour rest period, under pressure. For the influent challenge, general test water was spiked with appropriate contaminants to obtain influent challenge solutions with contaminant concentrations recommended by NSF/ANSI-42/53. Tests were conducted against:
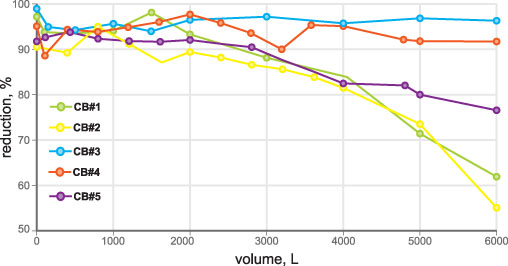
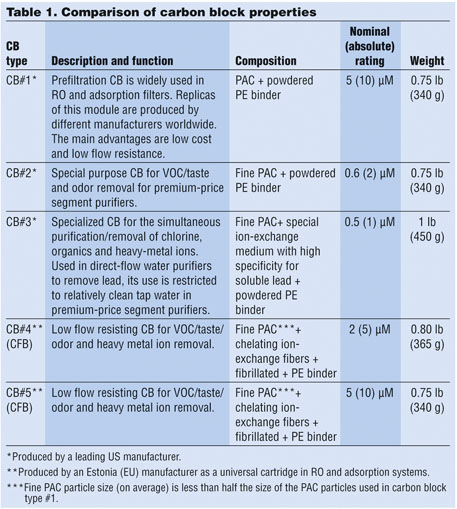
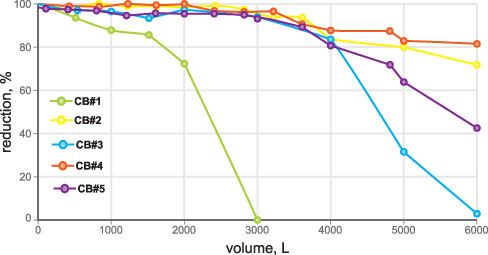
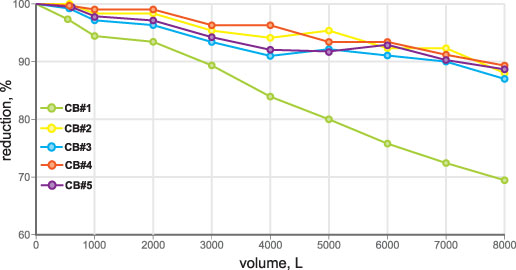
• VOC (chloroform as surrogate): 300 ± 30 ppb chloroform (threshold 15 ppb/95-percent reduction) (Figure 2)
• Total lead: 150 ± 15 ppb total lead (threshold 10 ppb/93.3 percent reduction) (Figure 3)
• Active chlorine: 2.0 ± 0.2 ppm (threshold 1.0 ppm/50 percent reduction) (Figure 4)
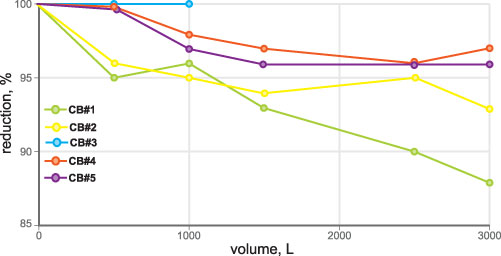
• Particle removal test: ISO 12103-1 A3 medium test dust (nominal 0-80 micron size) (Figure 5)
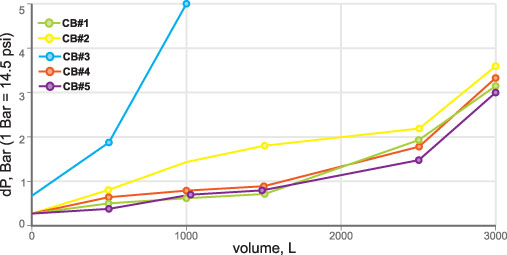
In the preparation procedure, before testing, filtration cartridges were flushed with tap water according to the operation manual requirements. Sampling points were 1, 100, 500 liters and each 500th liter (for VOC testing sampling points at 1, 100 and each 100th liter). Temperature, pH, turbidity and differential pressure (Figure 6) were also the subject of constant monitoring.
Results and discussion
In all carbon blocks, water flows around AC particles. Carbon particles are held together by sticky polymer particles. There are, typically, 12 to 22 percent w/w (polymer binder in the carbon block). Most AC particles are larger than the polymer binder particles, although most efficient carbon blocks contain significant quantities of AC dust, which provides explosive adsorption speed. Activated alumina powder in carbon block #3 is responsible for heavy-metals (Pb) removal capacity.
Because of the tortuous water path and generally hydrophobic nature of the carbon blocks, dirt particles plug up both the porous carbon block matrix pores and its outer surface, leading to the premature end of the carbon block’s useful life (versus its theoretical adsorption capacity).
Carbon fiber blocks (CFB) 4 and 5 also contain hydrophilic chelating (selective affinity toward heavy-metal ions) ion-exchange fibers 10 to 20 microns in diameter (Figure 7). The fibers are much longer than the AC and binder particles and provide uninterrupted flow paths matrix for the water.

The major difference between the same diameter hydrophilic fibers and particles is that an adsorption bed composed of fibers offers negligible flow resistance to fluids; the same adsorption bed composed of the same diameter particles has high-flow resistance. Water likes to flow along the hydrophilic fibers and its flow is also assisted by the capillary suction.
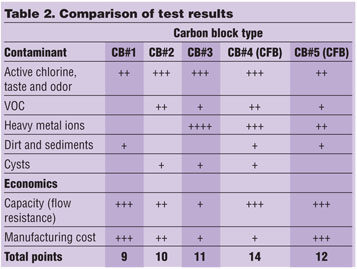 Carbon fiber blocks allow using PAC with a much smaller particle size as compared to standard carbon blocks. Powdered activated carbon provides superior organics removal properties while chelating ion-exchange fibers (chelation-specific irreversible binding of heavy-metal ions via complex formation) provide superior removal of heavy metals.
Carbon fiber blocks allow using PAC with a much smaller particle size as compared to standard carbon blocks. Powdered activated carbon provides superior organics removal properties while chelating ion-exchange fibers (chelation-specific irreversible binding of heavy-metal ions via complex formation) provide superior removal of heavy metals.
Conclusion
Carbon blocks can replace GAC in POU applications due to their superior kinetics and lower flow resistance. In turn, AC fiber blocks were found to be superior in the combination of the adsorption capacities and flow resistance as compared to the other carbon blocks.
About the authors
° Alexey Aksenov is Senior Research Chemist of WestAqua Ou, Aquaphor Group, Narva Estonia. He is a graduate of the St.Petersburg State Institute of Technology, Electrochemistry Department. Askenov can be reached at [email protected].
° Alexander Mitilineos, Head of R&D Chemistry Dept. of WestAqua Ou, Aquaphor Group, Narva Estonia, is a graduate of the St.Peterburg State University, Inorganic Chemistry Department. He can be reached at [email protected].
° Alexander Polyakov, corresponding author, is Vice President, Technical Sales of WestAqua Ou, Aquaphor Group, Narva Estonia. He is also a Graduate of the Leningrad State University Physical Chemistry Department’s PhD program. Dr. Polyakov can be reached at [email protected].
° Joseph L. Shmidt, CEO of Aquaphor Group, was also co-Founder of Membrex, Inc. (now a part of Osmonics, GE Water). He earned his PhD and MS Degrees in chemical engineering from Columbia University, NYC. Dr. Shmidt can be reached at [email protected].





