Numerous articles have been and continue to be written about the current methods for removing arsenic from drinking water. It is not the purpose of this article to
rewrite what has already been written but rather to introduce a new material and method for removing arsenic. This article will present the existing technologies in brief format, followed by the new methodology cerium oxide, a derivative of the rare earth metal cerium, then explain how each method fits in the industry.
Arsenic occurs in groundwater in two main species: trivalent and pentavalent. The level of toxicity and the complexity of removal depend upon which of these two forms is found in the water supply. Arsenite (trivalent arsenic) is significantly harder to remove due to its neutral charge and is only responsive to two of the four adsorption technologies presented. Having a negative charge, arsenate (pentavalent arsenic) is responsive to all of the arsenic adsorption media.
Currently, the three most common media for arsenic removal are ion exchange resin, activated alumina and ferric oxide or hydroxide. The newcomer, tetravalent cerium as cerium oxide, has had little press in potable drinking water applications due to the lack of commercially available delivery systems and lack of NSF approval, although trivalent cerium, as CeCl3, is already being used in the wastewater treatment industry. This product has tested very well for removal of phosphorous and arsenic, as well as many other contaminants. Until now, neither form of cerium oxide has been successfully applied in potable water treatment. CeO2 is commercially available as a 7-micron particle, which is ineffective in standard delivery systems due to high-pressure drop. A US-based R&D/manufacturing company has successfully created an effective CeO2 product that is WQA tested and certified to NSF/ANSI 61. Each of the aforementioned media have their strengths and weaknesses and each has an application that matches its abilities. The question is, which one best suits your client’s needs?
Ion exchange (IEx) resins
Ion exchange resins can be manu- factured to be the most selective of the adsorbents; however, the technology requires a charge for the purification to take place. Therefore, it is only functional with arsenate (V) unless an additional oxidizing agent is present. Ion exchange resins have the longest lifecycle of the adsorbents because they are easily regenerated; however, regulations on discharge water amounts and salinity levels are making this process more difficult. Anion resins are sensitive to sulfate and nitrate; these contaminants should be treated prior to arsenic removal. The process of using anion resin for arsenic treatment will also raise the level of chlorides in effluent water. Though re- generating ion exchange systems has a place in arsenic treatment, single-use virgin resin cartridges have too small a capacity to be useful. Ion exchange resin does not pass the toxicity characteristic leaching procedure (TCLP) for waste disposal.
Activated alumina (AA)
Activated alumina is likely the least expensive medium, but the total cost per milligram of arsenic removal is the highest. AA is pH sensitive and operates best at a range of 6-7.0. It is also the least selective of the adsorbents. Removal performance of AA is dramatically reduced in the presence of competing ions, such as chloride, fluoride, sulfate and phosphate. AA is also selective to arsenate and ineffective for arsenite. POU AA cartridges are very popular in the marketplace for general fluoride and arsenic reduction. Most activated alumina media will pass the TCLP for waste disposal.
Ferric oxide and hydroxide (GFO, GFH)
Granular ferric oxide and hydroxide media are probably the least expensive to operate on a large scale. GFO is less pH sensitive than activated alumina but still has its limitations. The optimal operating range for GFO is 6-8.5, with lower ranges yielding the highest capacities. GFO is the first of the adsorbents to handle both arsenite and arsenate, though in higher flowrates, an added oxidizer is recommended to aid in arsenite removal. GFO media are currently the most commonly used to focus on arsenic removal, in both larger POE systems and POU cartridge- style systems. The highest quality GFO media removes an average of 4 mg/cubic inch of media volume at a suggested 0.5 gpm (1.8 liter/min) maximum flowrate for most cartridges. GFO media is able to pass the TCLP for waste disposal.
Reticulated cerium oxide (5D)
5D cerium oxide media is the newest adsorbent for arsenic removal. It is comparable in price to GFO media cartridges but has a capacity of 6-8 mg/cubic inch. 5D cartridges also have an operating flowrate of 1 gpm (3.7 liter/min) for a 2.5- x 10-inch (6.35 x 25.4-cm) cartridge and 4 gpm for a 4.5- x 20-inch (11.43 x 50.8-cm) cartridge. This is a significant increase in flowrate and capacity in comparison to GFO cartridges. Current 5D industrial systems, however, are less economical than GFO in industrial systems. 5D media have tested very well in pressurized cartridge applications and excel in gravity-flow applications, making them a good fit for smaller POU/POE systems. The medium is a natural adsorbent, so there is no foreseen issue with TCLP testing, though that process is currently underway.
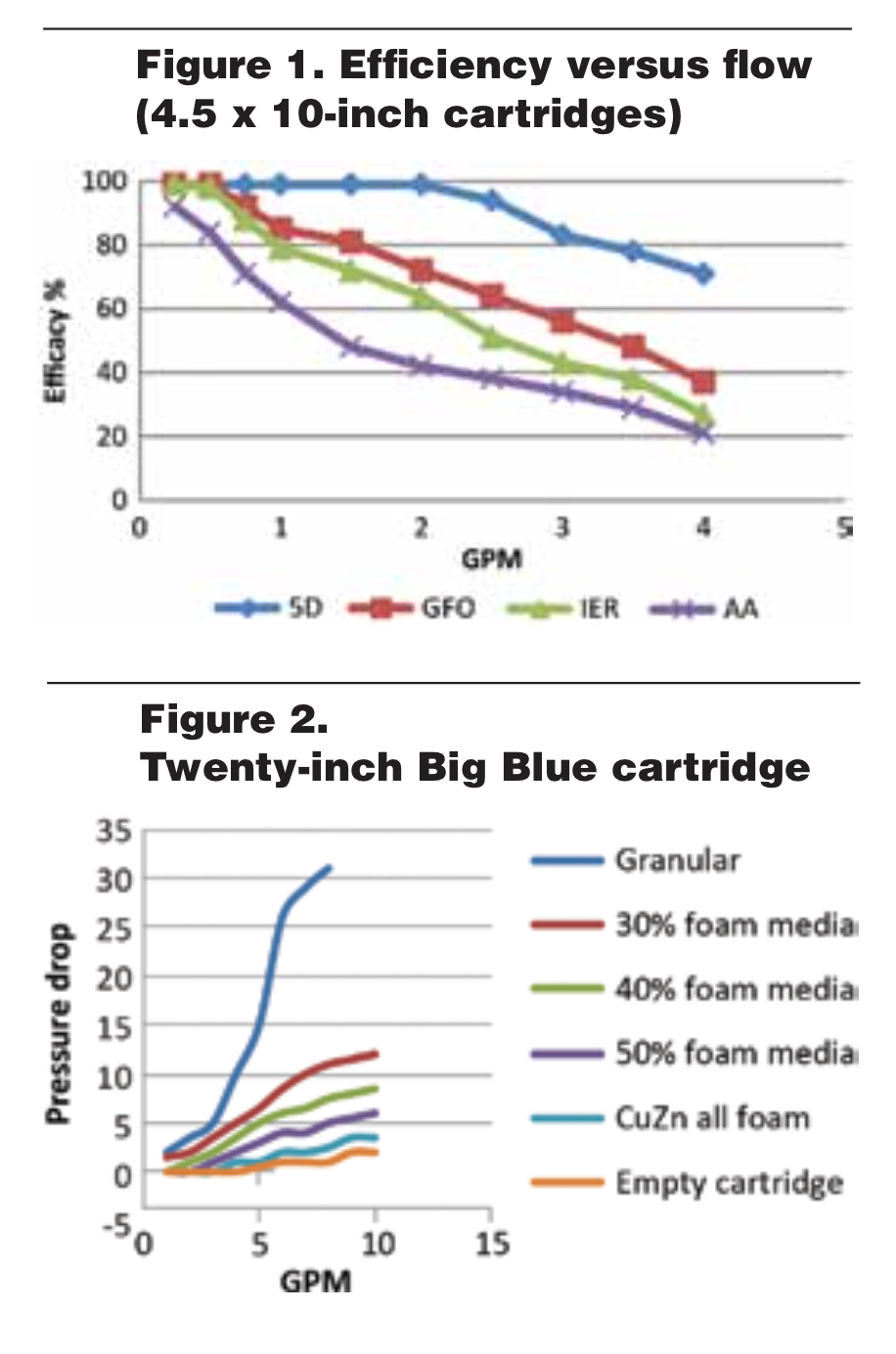
Reticulated-media performance is a direct result of chemical kinetics and the Collision Theory (see Figure 1). This is accomplished by the size of the particle and the reticulated structure, which causes a tortuous path for the influent gas or liquid solution. The law of mass action states that the speed of a chemical reaction is proportional to the quantity of the reacting substances. In the case of reticulated media, it is the quantity of readily available surface area. Collision Theory states that the more collisions created, the higher the percentage of chemical reactions. The reticulated structure assures the influent stream will see a maximum number of collisions with the most efficient particle size possible. It is because of the reticulated structure that 5D media can be used in gravity-flow applications, as well as create little pounds-per-square-inch differential (PSID) in pressurized applications (see Figure 2). It can provide developing countries with a fast flow, small footprint, easily operated solution to safe, arsenic-free, potable water.
Conclusion
When sourcing media to deal with arsenic issues, there are many variables to consider. The choice of filtration material will be crucial in handling arsenic problems in an efficient way. Though many media are available for arsenic removal and reduction, two in particular stand out as leading technologies: GFO media for larger industrial potable waters and 5D media for smaller POU/POE applications. Continuing to research new, emerging technologies will go a long way toward a successful, efficient and profitable arsenic removal program.
Writing: Bob Brooks
About the author
Bob Brooks is a partner and Chief Engineer of Research and Development at Foamulations LLC. He has a background in engineering and has been in the water treatment business for over 14 years. When Brooks is not helping a customer to develop their new applications, he is researching new media and new ways to use reticulated foam products. He is also an accomplished bass player and performs regularly in various venues. Brooks can be reached via email, [email protected]
About the company
Foamulations LLC is an R&D Engineering company as well as a manufacturer of reticulated foam delivery systems for new and exist- ing media. Foamulations supports its products with a thorough testing protocol and now has multiple WQA certifications under the parent company Filter Foam Technologies. Visit www.foamulations.biz
About the product
5D media is a reticulated delivery system that typically boosts efficacy and increases capacity. The reticulated foam structure prevails in low- pressure/gravity-flow situations as well as high-flow situations where a low PSID is required. The porous nature and large surface area of the reticulated structure brings versatility and breakthrough technology together in both liquid and gas phase filtration.





