The world is inexorably moving in the direction of water conservation and reuse. Media coverage of extreme water shortages, disease and chemical contamination events has raised public awareness to unprecedented levels. Increased regulatory activity, both in the area of contaminant discharge and total water usage is causing commercial and industrial water users to look at ways to conserve and reclaim their wastewater. Certain areas of the US have already initiated regulations limiting or eliminating the installation and usage of self-regenerating ion exchange systems, primarily due to the high levels of chlorides being discharged into our limited water supplies.
A new, patented treatment system (US Patent #:8,580,118) has been developed to recover most of the brine from the water softening regeneration process. It utilizes nanofiltration (NF) tech- nology to separate hardness ions from brine solution, discharging the hardness to the drain and returning recovered sodium/potassium chloride to the brine tank for reuse. Production-sized units have been manufactured and testing is ongoing. The preliminary results show that approximately 75 to 85 percent of the brine can be recovered. This provides:
- Significant cost savings for the end user. Example: a Mexican restaurant located in Las Vegas, NV spent $800/ month on salt for their two five-cubic-foot water softeners. Conservatively, a 75-percent reduction would result in a $600/month salt savings alone.
- Less maintenance required for the end user, reducing time spent adding salt to the water softener by 80 percent (+/- 5 percent)
- A dramatic reduction of salt (monovalent ions), being added to our water supplies
- A new potential to get water softeners reapproved in brine-restricted areas
- Approximately 35 to 40-percent reduction in water used during the regeneration process
This process operates in a batch mode: the slow rinse and part of the backwash water is directed to a separate tank (Re- claim Tank) for treatment by the NF membrane that directs the concentrate to drain and permeate to the brine tank. Following are the steps involved:
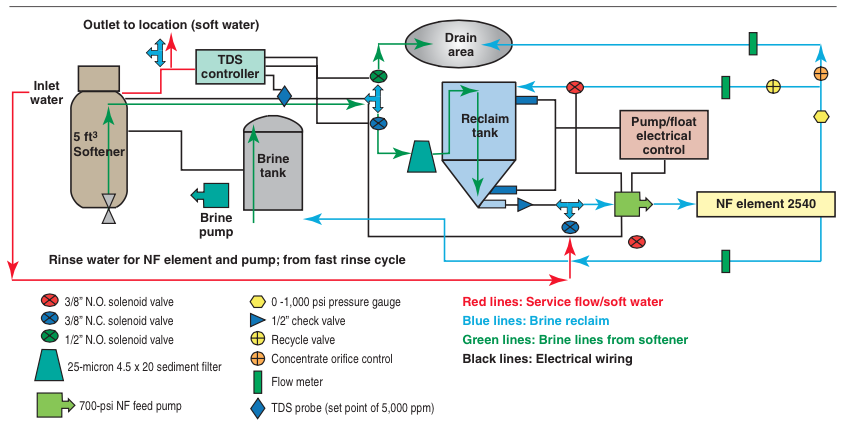
1) Upon initial start-up, the brine tank is filled to the appropriate level with tap water (or softened water). After the first regeneration, the system is configured to add the appropriate levels of water back into the brine tank, thus mostly eliminating the ‘fill cycle’ of the water softener. (Some fill may be required to account for water lost from evaporation).
2) Once the ion exchange system begins, the brine-draw/ slow-rinse cycle, the water passes through a conductivity controller, wherein the conductivity of the brine-draw/slow- rinse water is monitored. When the conductivity reaches the appropriate levels, the brine water is redirected into the Reclaim
Tank. In a five-cubic-foot system, the amount of water collected is approximately 95 gallons (360 liters).
3) The Reclaim Tank contains two level switches. Once the brine water level (with high concentrations of NaCl or KCl) reaches the top level control, a pump is activated through the electronics. The pump draws water from this tank and directs the brine water through the specifically configured NF membrane element at approximately 700 psi. The majority of monovalent ions (75 to 85 percent) are redirected back into the ion exchange system’s brine tank, while the multivalent ions are routed to the drain.*
4) The system is configured so that the amount of brine water directed back into the ion exchange brine tank is equal to the amount of water introduced during the fill cycle.
5) Once water in the Reclaim Tank drops down to the lower float control, the system’s electronics shut down the pump.
6) After the pump has shut off, the electronics then activate a set of solenoid valves that allow tap water (or softened water) to flush through the pump and NF element, minimizing pump corrosion and membrane fouling from the high salinity water.
7) At the conclusion of the flushing cycle, the system shuts down and won’t be active again until the ion exchange system goes through its next regeneration process.
An illustration of the overall system configuration is shown above. To reduce the water usage as much as possible, the eductor is replaced with a brine pump and the freeboard in the resin tank is reduced. These minor changes allow this system to retrofit existing softener units.
Conclusion
According to a recent report from NJ-based Baytel Associates (www.baytel.org): “The market for commercial water softeners in the USA and Canada came to about a $250,000,000 in 2014 and is projected to resume healthy annual growth rates of 6% or more by 2017.” These data indicate the potential for new softener installations in the commercial/industrial arena. When factoring in the aforementioned benefits of the system, the market for this technology, as either an add-on feature to existing equipment or a complete new system sale, is remarkable.
* This 75 to 85 percent calculation is based on a straight comparison between the sodium chloride (converted to pounds of NaCl) drawn from the water softening brine tank subtracted from the total amount of sodium chloride (also converted to pounds of NaCl). The amount of sodium used to regenerate the resin bed is a part of the 15 to 25 percent that is lost. On average, it took 16.7 pounds of sodium chloride to regenerate the five-cubic foot-softener, and between 13.5 to 14.2 pounds of NaCl were reclaimed and reintroduced to the softening brine tank.





