By C.F. ‘Chubb’ Michaud, CWS-VI
Ion exchange is versatile
When you think about water treatment processes that can remove the dissolved salts from feed streams, there are many options. Ion exchange, reverse osmosis, distillation or electro-deionization (EDI) are all possibilities. But what if you don’t want to remove everything from the water? What if you want to leave something behind for economic or health reasons? What if you want to go between the lines and remove only a particular ion from a mix? Ion exchange has the flexibility to do all of these.
Ion exchange works because of ion selectivity, mass action and system equilibrium. Simply put, if a substance is in the water (ionic) and is not on the resin, it will go onto the resin. If it is on the resin and not in the water, it will go into the water. This is what contributes to the all-familiar hardness leakage in a water softener when it is not fully regenerated, and what allows us to remove sodium with a potassium-regenerated water softener (or remove potassium with a sodium-regenerated system). It is why we can soften seawater and use seawater as a regenerant (albeit, not at the same time).
(Courtesy of Dow Chemical)
There are four basic types of ion exchange resin and a blend of these resins used in everyday demineralization processes. This article will discuss those types of resins and review how the water composition changes when passing through beds of those resins. We are not including chelating resins, boron-selective or perchlorate-selective resins, but will focus on the most widely used:
1. Strong-acid cation (SAC)
2. Weak-acid cation (WAC)
3. Strong-base anion (SBA)
4. Weak-base anion (WBA)
5. Mixed-bed DI
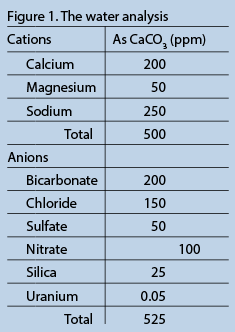
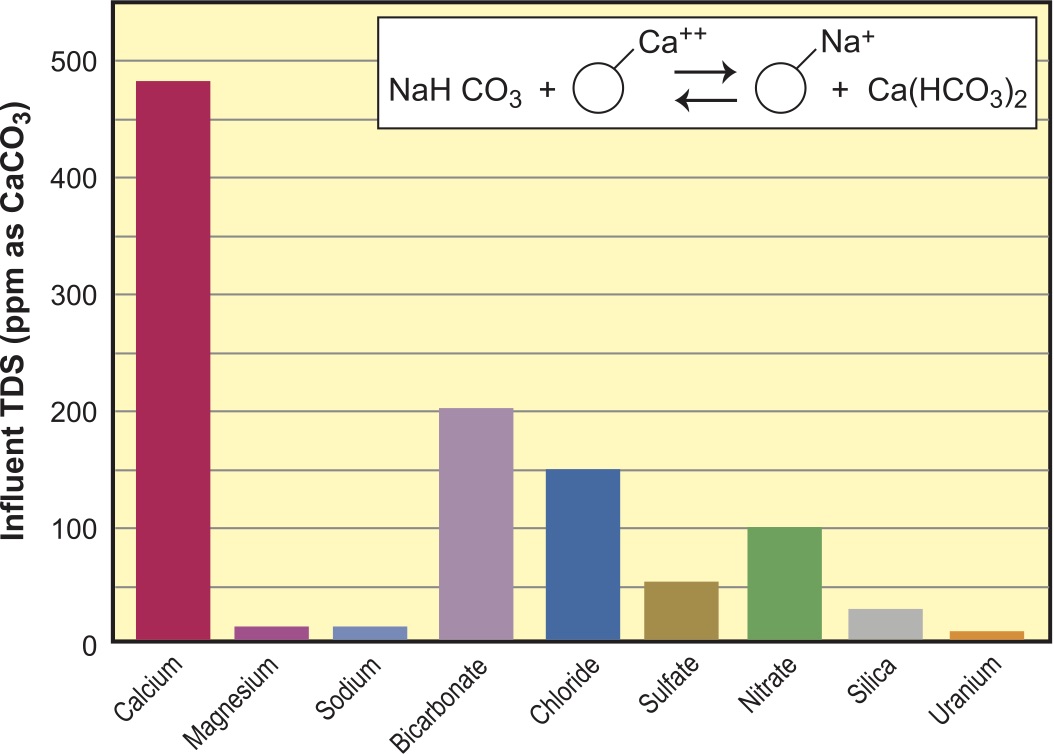
For simplicity and purposes of comparison, we will use the same water analysis for each resin reaction and follow the before and after in graphs. The water we have chosen is a moderately hard and moderate TDS (total dissolved solids) water, typical of many parts of the country. We have added a farm flavor with high nitrates, and to spice things up we have uranium at just above the US EPA maximum contaminant level (MCL). You will note that the water analysis is balanced to 500 ppm as CaCO3 for the anion loading, and that silica is an add on (Figure 1). Since ion exchange resin selection is partly driven by the water analysis, I would be remiss without a quick review of the relevant chemistry.
Ion exchange is a natural phenomenon in nature. It is the mechanism by which soil takes up certain nutrients and plays an important role in the growth of most living things. Soil contains zeolites, which are strong-acid, inorganic mineral lattices that can act as softeners. If soil is high in divalent calcium (Ca+2) and magnesium (Mg+2), it will shrink and provide good drainage. If soil is fed a steady diet of softened water, however, it will exchange sodium (Na+1) in the water for hardness on the zeolite and swell, causing poor drainage and choking off the roots of the plant. This same phenomenon may also interfere with septic drainfields if an online softener is not allowed to discharge to the septic to replenish the hardness in the soil.
Ion exchange selectivity
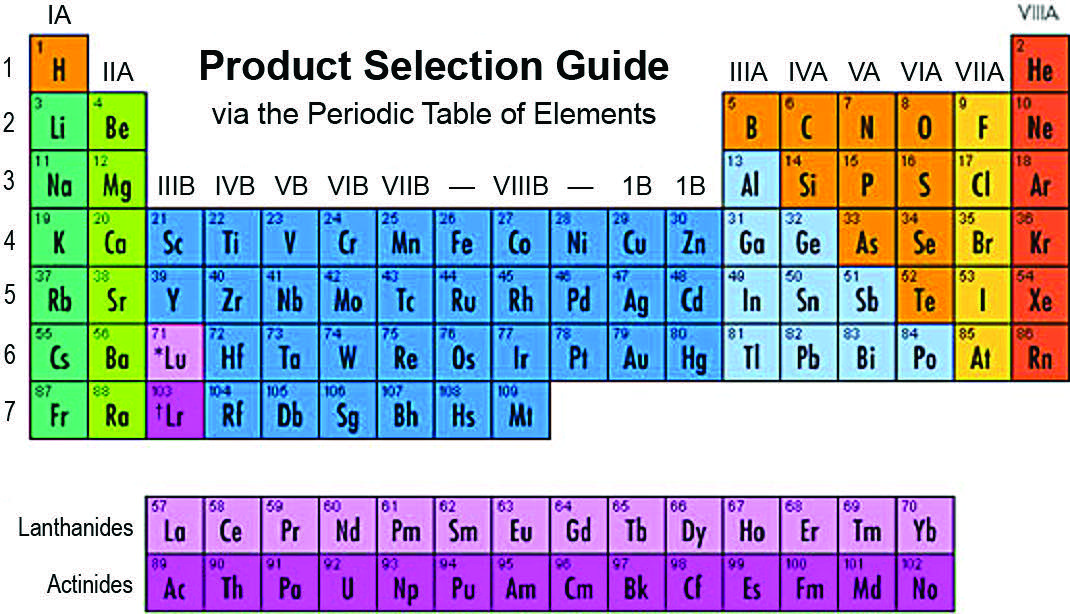
Ion exchange works, in part, by selectivity. That is, ions with higher charge potential (valence) will usually replace those with lower strength charges. If we look at the Periodic Table of the Elements (Figure 2) in Column IA containing Na+1 and potassium (K+1), we see that selectivity increases as the molecular weight (MW) increases (K+1> Na+1> Li+1) going down the column. Column IIA, containing divalent hardness Mg+2 and Ca+2, as well as strontium (Sr+2), barium (Ba+2) and radium (Ra+2), all have a higher charge, and MW also increases as you go down the column. The selectivity for any of the ions in IIA is higher than those in IA, which is why a softener works and why you can use a softener to remove radium even beyond the hardness break (Ra+2 will replace Ca+2 on the resin).
Moving to Column IIIA, we see aluminum (Al+3). The trivalent charge of aluminum makes it very tightly held by a softener resin, and it may actually foul the resin because it becomes very difficult to remove unless regenerated with very high doses of salt at higher concentration (i.e., 15 percent). Generally, the farther down the column and the farther to the right, the more selective the ion over sodium. Those elements that form anions have a similar selectivity (I-1 > Br-1 > Cl-1 > F-1, Column VIIA) but it gets more complicated when the elements combine with oxygen. The following will help put a value on selectivity.
Strong-acid cation (SAC 8-percent crosslinked) selectivity

• Trivalent
Lanthanum > cerium > chromium
• Divalent
Barium > lead > strontium > calcium > manganese > beryllium > nickel > cadmium > copper > cobalt > zinc > magnesium
• Monovalent
Silver > cesium > rubidium > potassium > ammonium > sodium > hydrogen > lithium
Sodium is highlighted to show its relative position. As with any rule of thumb, there are exceptions. Barium is more selective than lanthanum. Lead is more selective than chromium (Cr+3—Cr+6 is an anion specie). Silver is more selective than chromium and strontium. In fact, if you increase the crosslinking (the divinyl benzene content) to 10 percent for SAC resin, it becomes more selective for silver than any other ion listed here.
Strong-base anion (SBA Type I) selectivity
Perchlorate > uranyl carbonate > citrate > iodide > bisulfate > nitrate > bromide > nitrite > cyanide > bisulfite > bromate > chloride > hydroxide > bicarbonate > acid phosphate > fluoride
Chloride is highlighted to show its relative position; the above is for a Type I SBA. Type IIs and weak-base anion have a slightly different order, which makes resin choice of prime importance when addressing different streams. Sulfate, for instance is preferred about 1.3:1 over nitrate with a Type I, whereas it is about 2:1 with a Type II. This makes a Type I less likely to dump nitrate towards exhaustion. Anion exchangers can have multiple chemical functionalities, which can shift the selectivity from one ion to another for a specific task. Nitrate selective resins, for instance, will prefer nitrate over sulfate and may be preferred for safety in residential installations.
Chemistry 101
The simplest reaction we learned in high school chemistry was that of neutralization. When you mix an acid and a base, you form a salt and water. In the following example, we are using hydrochloric acid (HCl) and caustic (NaOH) but the same reaction holds true for any acid and base.
- HCl + NaOH → NaCl + H + OH (H20)
- acid base salt water
Since the products of reaction are soluble, there is no way to use this type of reaction to separate the salt and the water that is formed. If we had a form of acid and base that were insoluble, however, the salt and water could be separated. This is exactly what ion exchange does.
Ion exchange reactions
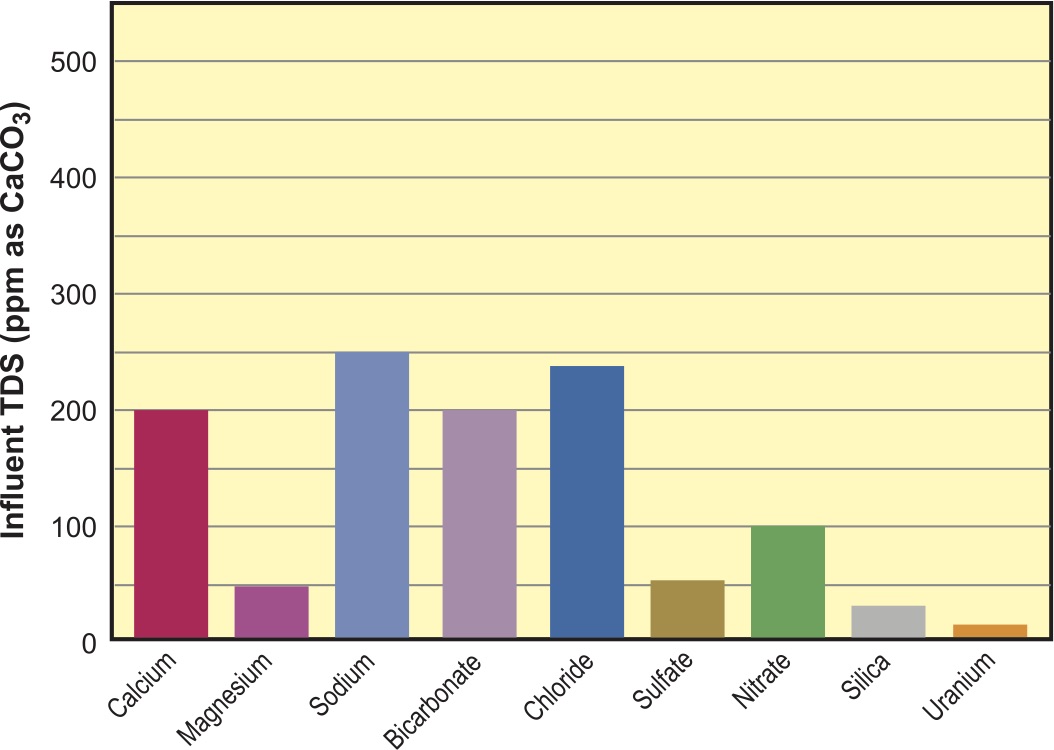
In Figure 1, we see the water analysis for the feedwater we will use for the rest of this article. Figure 3 is the bar graph representing this analysis.
The first and most common use of ion exchange is the water softener. This reaction is shown in Figure 4 along with the resulting bar graph for the change in the water composition.
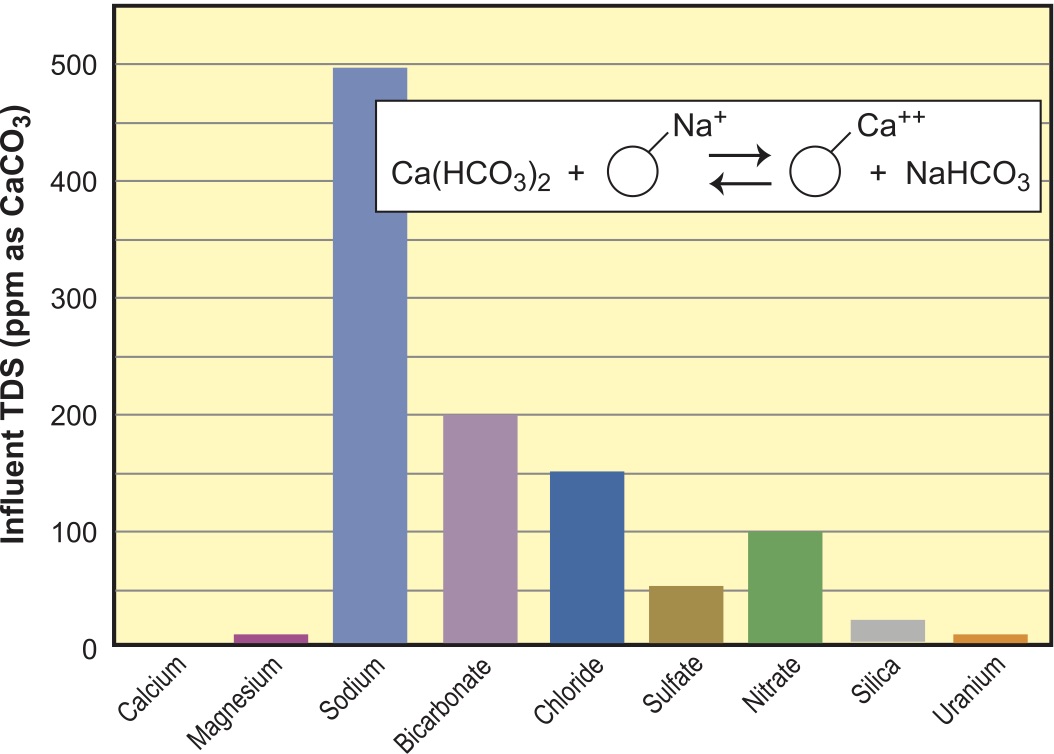
Here, the softener has exchanged sodium for the calcium and magnesium hardness. You will note that the Ca has been completely removed and there is a small amount of Mg in the product water representing leakage, which is normal. Depending upon the level of salt used for the regeneration, leakage may be < 1 ppm or a few ppm. Sodium is increased by the same amount that hardness has been decreased. TDS does not change.
Referring to the earlier selectivity series, we can note that a softener is capable of exchanging just about every cation above sodium in the series, including such ions as lead and copper and a host of heavy metals. Softeners can often be used for cleaning up plating rinses before disposal. One should use softened water as the rinse water makeup so the second softener will only see the sodium in the water plus the metals from plating rinses. None of the anions have changed and the TDS of the stream remains the same as the raw water.
A second common use for ion exchange is when we use a salt-regenerated anion to reduce alkalinity (see Figure 5). Here, the exchange is for the chloride on the anion exchanger, and we are showing the bar graph for the same water that has already been softened.
Note the small amount of alkalinity leakage. Since the resin actually prefers chloride over alkalinity, we usually run into some low-level breakthrough on alkalinity in the range of 10 percent of the total. At first, it will remove all of it, but we run past the initial alkalinity break for reasons of economy. Note also that sulfate, nitrate and uranium have also been removed and replaced with chloride. The increase in the Cl level represents the total of the other ions that have been reduced. Silica is not reduced because silica is so poorly ionized that it really doesn’t act as an exchangeable ion, other than with an anion resin in the hydroxide (OH–) form (more later under deionizers).
The deakalizer can utilize either Type I or Type II anion resins and can be used for the reduction of sulfate, nitrate, hexavalent chromium and any of the other anions shown in the selectivity series above, including selenium, arsenic and even fluoride. Note that fluoride falls below chloride in selectivity, so capacities will be low and should be followed by a fluoride-selective adsorber, such as activated alumina. Since the dealkalizer removes the bicarbonate ion, it reduces the pH of the treated water, which improves efficiency and capacity of the polishing fluoride filter. A seldom-used realization for the salt regenerated SBA dealkalizer is that of anion softening. The bicarbonate ion is removed and the resulting water will not form scale when heated, even without softening the water first. The resulting salts are all chlorides, which will have very high solubility. This can be done with just the SBA for scale prevention in boiler feeds.
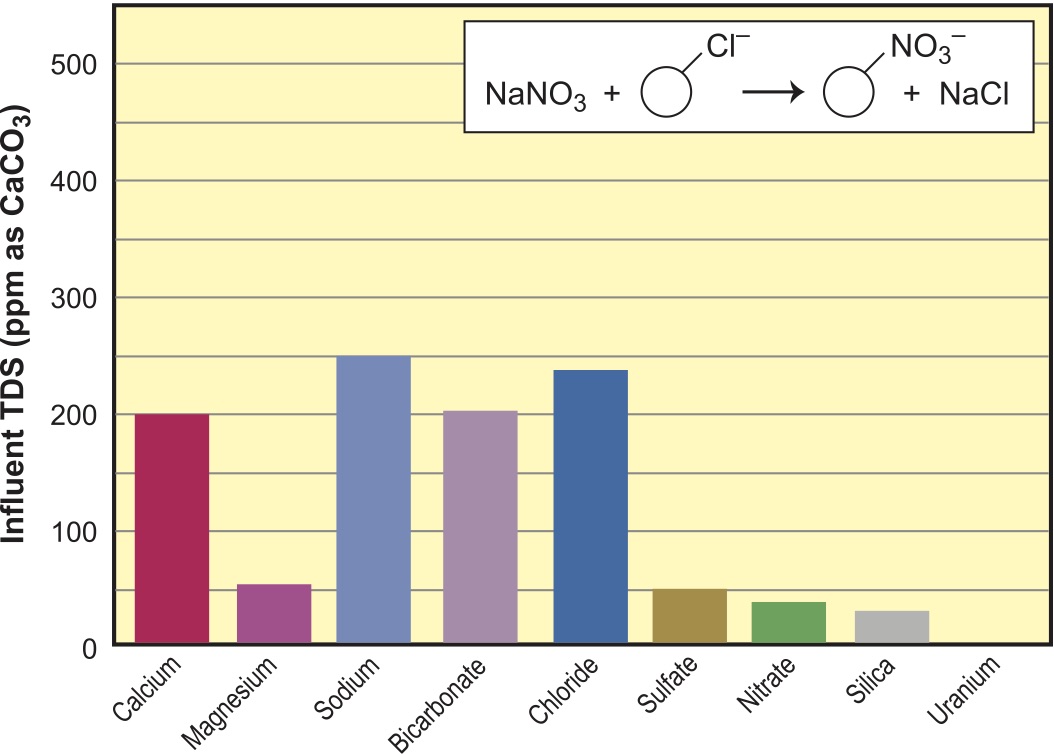
Figure 6 shows a graph of treated water using a nitrate-selective resin without softening. At first, the resin removes all anions except silica, replacing them with chloride, and is then followed by a breakthrough on alkalinity. As the resin continues to load up, it starts to break on sulfate while still removing the majority of the nitrate. Eventually, near the end of the run, the nitrate will start to break and be allowed to run up to the limit before terminating the run. The resin continues to remove uranium because of the very high selectivity for that ion. By carefully selecting the resin, one can design systems to remove multiple contaminants with a single-resin bed. Such a system was covered in a previous article2 in the city of McCook, NB, utilizing a single-anion resin bed to remove arsenic, uranium and nitrate at the same time. One needs a very, very good and reliable water analysis to attempt this, however.
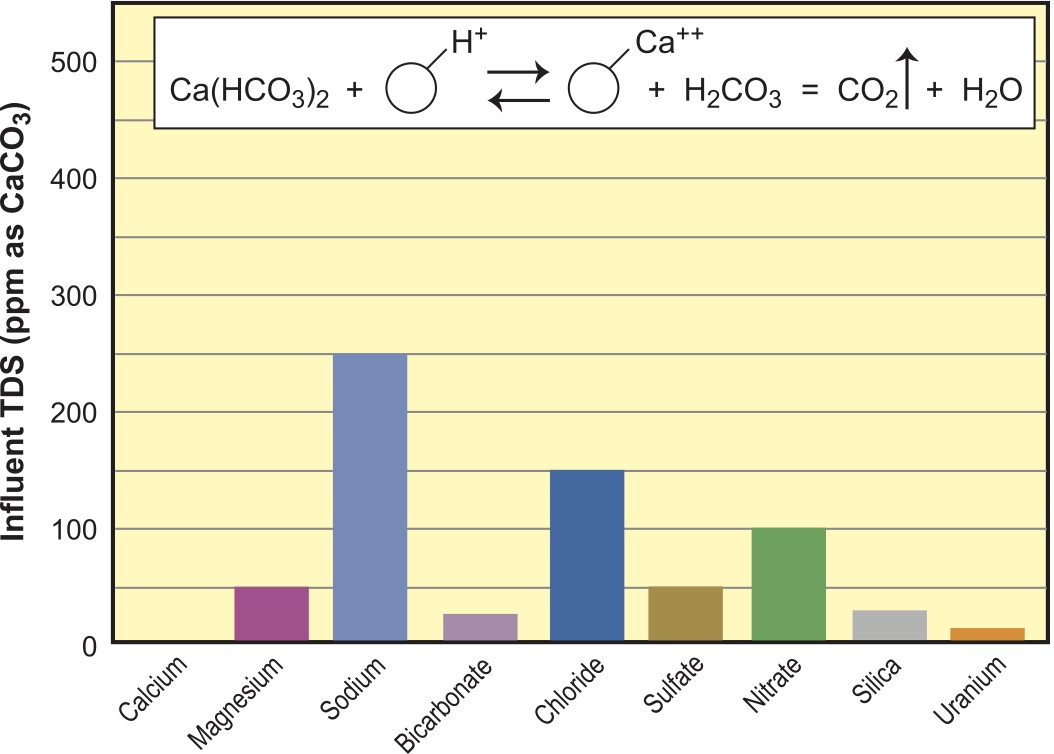
If faced with a water requiring both softening and dealkalizing with the right kind of water, it is possible to accomplish both with a single resin and reduce TDS at the same time. This is managed with a weak-acid cation exchanger run in the hydrogen (H+) form. This resin does not split a neutral salt (such as CaCl2) but can react with an alkaline salt (Ca(HCO3)2) and replace Ca hardness with H+ in the stream. This in turn drops pH of the stream, converting the remaining bicarbonate (HCO3)-1 to carbon dioxide gas (CO2) and water (H2O), as seen in the imbedded reaction in Figure 7. It is necessary to bubble off the gas created to neutralize the acidic stream, ridding it of carbonic acid. Note the water composition is lower in TDS by the 200 ppm of Ca removed and most of the 200 ppm of alkalinity. Na and Cl did not increase, so we now have a 300-ppm water, down from 500 ppm in the feed.
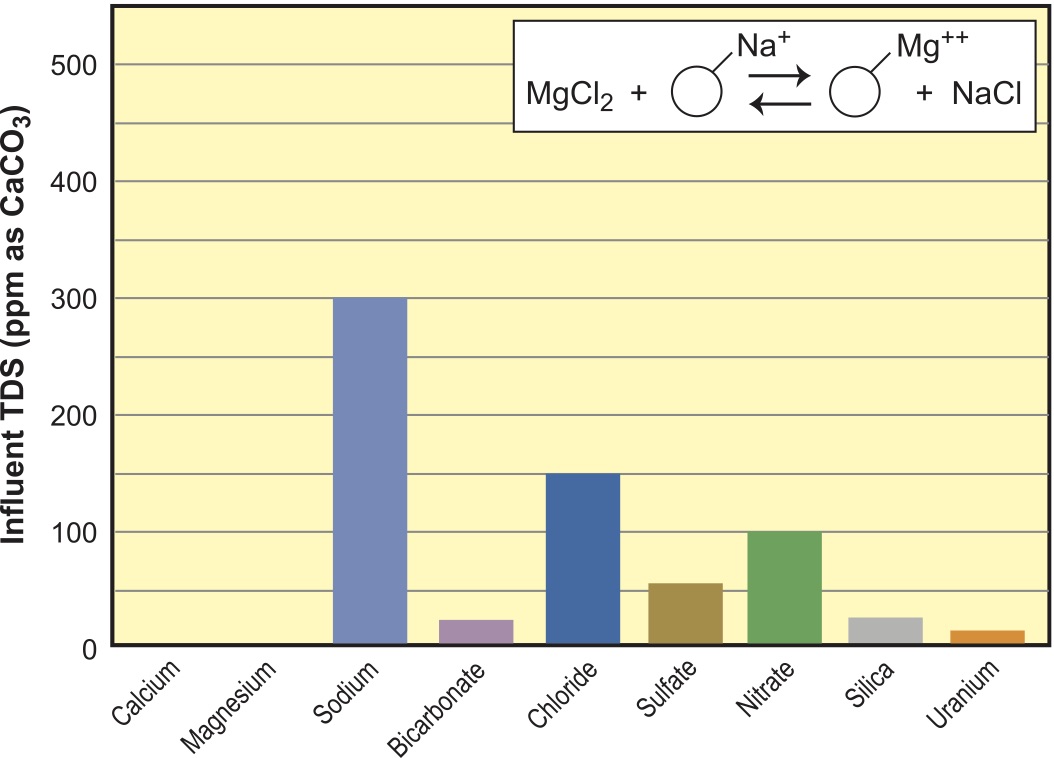
WAC resins are underutilized in the deionization world. They have nearly two times the capacity of SACs and can be run in either the H+ or Na+ form, but cannot be regenerated directly into the Na+ form with NaCl (neutral salt). They must first be regenerated with acid to put them back into the H+ form, and then neutralized with caustic (NaOH) or soda ash (Na2CO3) to put the resin into the salt or Na+ form. WAC resins in Na+ form have the ability to soften water and remove heavy metals from streams with much higher TDS backgrounds than do SACs.3 In the Na+ form, the kinetics are very rapid; a relatively small bed can do a mighty big job on low levels of metals (such as copper or lead).
WAC resins can soften a stream up to the limits of the alkalinity. In other words, if the hardness is higher than the alkalinity, there will be a moderate residual of hardness left, as shown in Figure 7. What do we do to remove all of the hardness and still have the reduced TDS and alkalinity? We soften, as shown in Figure 8, using a conventional SAC unit. This water represents a vast improvement over a straight softener for feeding low-to-medium pressure boilers because it is not only softened, but low in alkalinity and reduced TDS.
Demineralized water (also known as DI water) is achieved when minerals have been removed. Not all minerals, however, have to be removed to give DI water great utility for car washes, bottling plants and many manufacturing and cleaning operations. Consider the setup of SAC in H+ form and WBA shown in Figure 9.
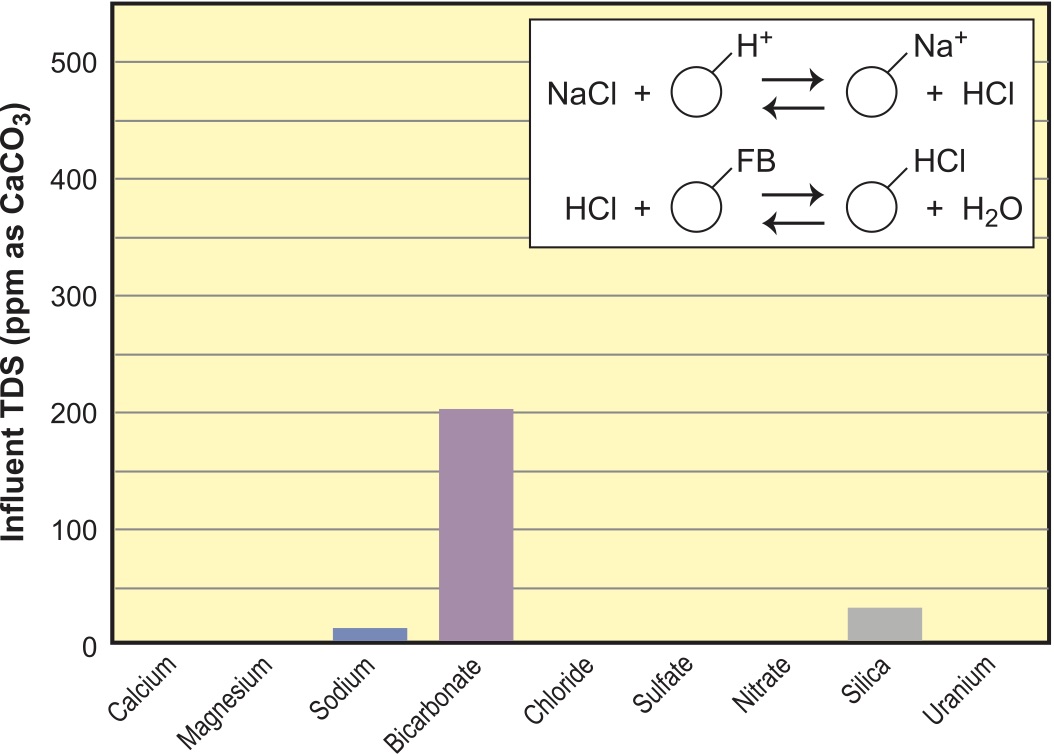
With the SAC run in H+ form, essentially all cations are removed and anions are converted to their acids. WBA resins, like their WAC cousins, do not split a neutral salt but will neutralize an acid (except for the weak acids of carbonic and silicic [shown]). As this water dries down, bicarbonate converts to CO2 gas and evaporates along with the water, as shown in Figure 10. This is the classic, spot-free car-wash rinse. If silica levels are 20 ppm or higher, there may be issues with severe spotting. In this case, you would need to go with compete deionization using SBA resins in a mixed bed. If you are using the water in some process where it is not evaporated and must be low in alkalinity, you can degas after the SAC, as illustrated by Figure 10.
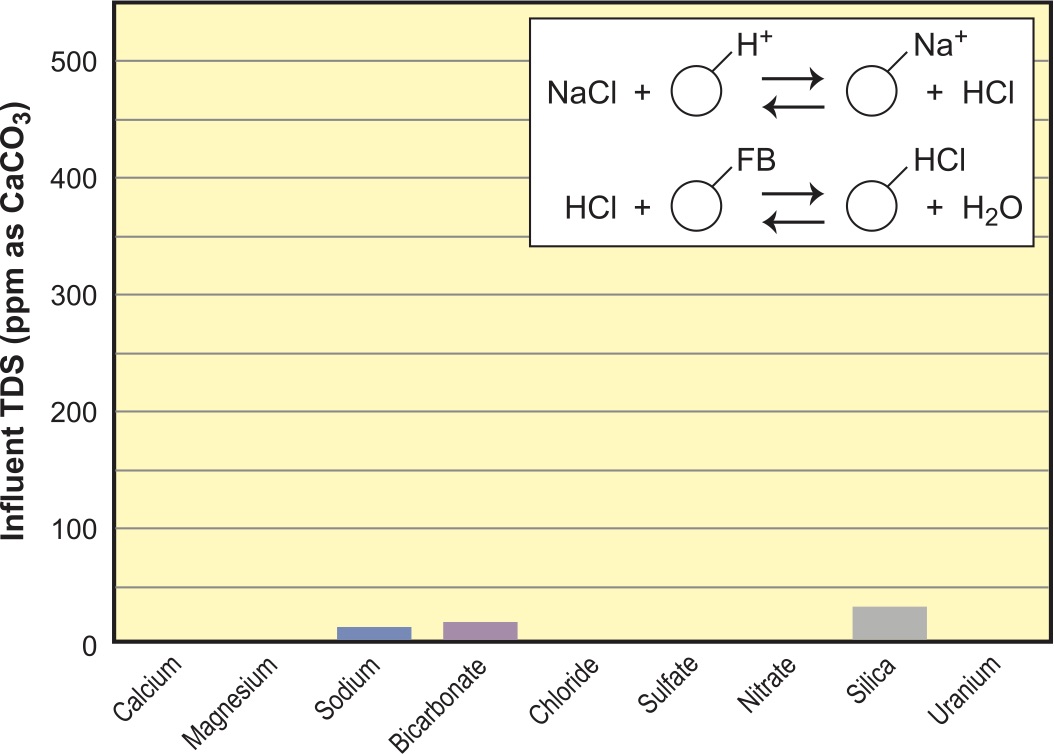
From here on out, we get into more serious demineralization where the goal is to strip the water of all conductive minerals, including silica. Silica in water is generally present as the weakly ionized silicic acid and can only be removed with the OH– form of SBA. For this purpose, we use a conventional two-bed DI setup with SAC in H+ form and SBA in OH– form. Residuals from this combination are a fractional ppm of sodium and ppb levels of silica (see Figure 11). You can generally anticipate Na leakage of about 0.5 percent of total cation feed with typical downflow levels of acid (7 to 9 lbs./cu. ft. or about 125 gm/L of acid/resin). In our example with 500 ppm of feed TDS, we would expect about 2 ppm of Na in the product stream. Using the same level of chemical for the SBA (Type I) and hot water (120°F or 50°C), we will see < 10 ppb of residual silica. The effluent shown in Figure 11 is basically a very dilute solution of NaOH with a pH in the range of 9 to 10. Note: a poor regeneration on the cation will lead to higher Na leakage and higher pH effluent, which will adversely affect silica leakage. Type II resins used in two-bed DI will have a higher capacity (by up to 30 percent) than will a Type I two-bed, but silica leakage will be ~100 ppb or higher.
In order to approach complete demineralization, it is necessary to polish out the remaining sodium; this is typically done with a mixed-bed polisher. It can be very efficiently done, however, by using a second SAC in H+ form as the polisher.4 Using conventional downflow regeneration, this system can produce up to 1 meg ohm (1 µS) water without the hassle of having to do mixed-bed regenerations. If one opts for counterflow regeneration, 10 meg ohm (0.1µS) water quality is achievable (see Figure 12).
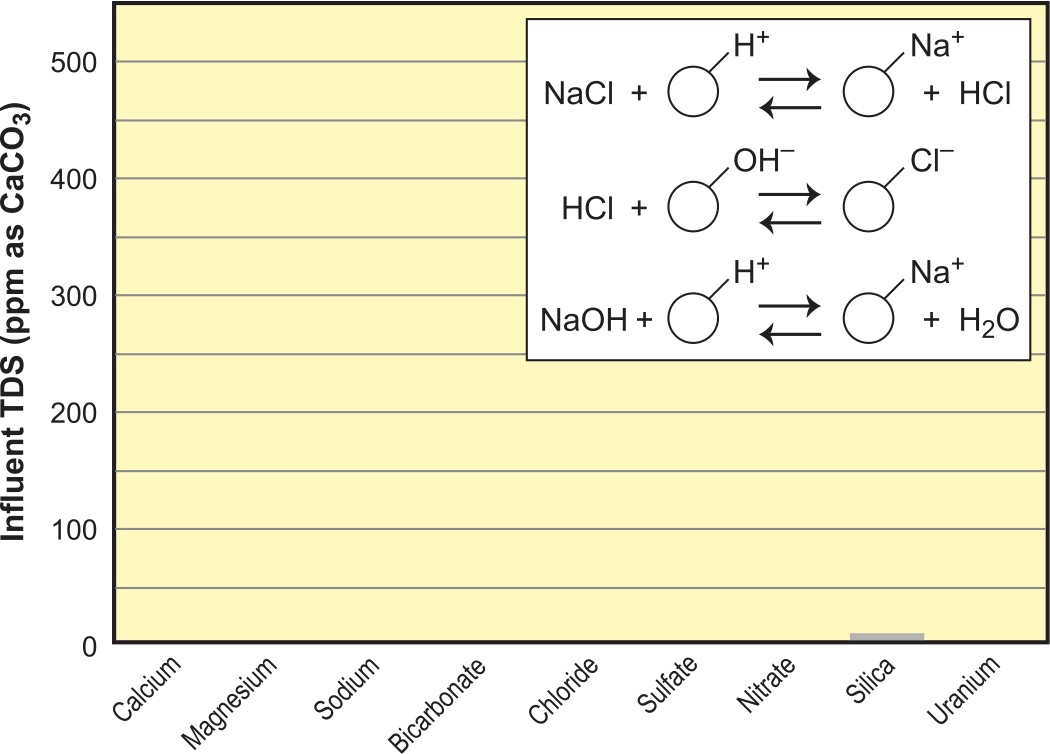
The ultimate in DI quality is achieved with mixed-bed resin. As shown in Figure 12, following a regular two-bed system with a SAC polisher will reduce the sodium leakage into the low, double-digit ppb range. The same would be true if the third bed was followed by a fourth bed, consisting of another SBA in OH– form. The silica residual would drop into the single-digit ppb levels as well. Now repeat this over and over with a near infinite series of SAC/SBA beds and you have mixed-bed quality.
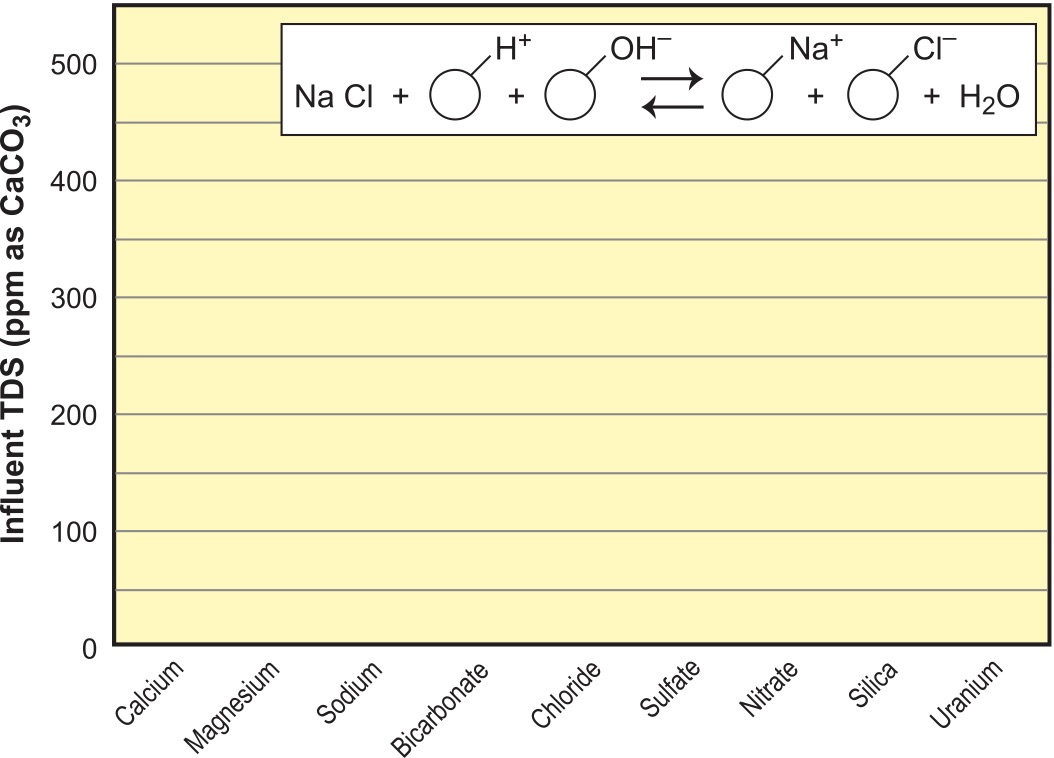
A mixed bed consists of an intimate mix of SAC and SBA resins in the same vessel, which will produce the ultimate in DI quality, as shown in Figure 13. The only drawback to mixed-bed DI systems is that mixed resin must be separated before it can be regenerated and reused. For portable exchange deionization (PEDI) operation, this requires a special separator that backwashes the resin and takes advantage of differences in density of the two resins to float the SBA to the top where it is transferred to a separate regeneration vessel. SAC is transferred from the bottom to its own regeneration vessel. After regenerating, the two resins are rinsed with DI water and transferred to a remix vessel where they are recombined by mixing with air. Fixed bed, mixed beds are regenerated in place with a rather complex distributor setup inside the tank that allows SBA to regenerate downflow after separation, and allows SAC to regenerate upflow simultaneously. After rinsing, they are remixed in place with air. Mixed-bed resin systems routinely produce 18-meg-ohm water (0.056-µS).
Disposable ion exchange systems
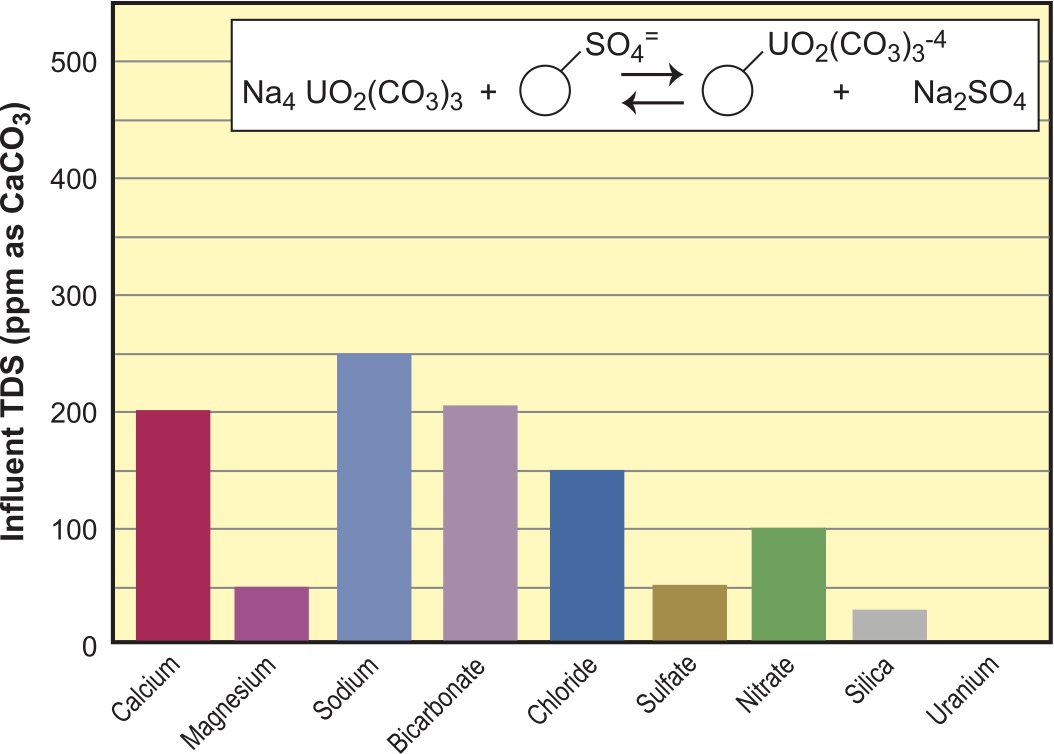
Certain ions are so highly selective for ion exchangers that they continue to run long beyond the normal ionic break for the usual ions found in feedstreams. Of note are perchlorate and uranyl ion (uranum). Often, these are the only contaminants in water that need to be addressed. Perchlorate is held so firmly that it cannot be regenerated off SBA resin, even with unlimited amounts of brine or caustic. Resin throughput capacities are in the million-gal./cu. ft. range (135,000 BVs). Another highly selective ion is uranium, usually present as an anion complex, shown in Figure 14 as the carbonate complex. Uraniun is so highly selective that it can be run to the point where the entire bed becomes highly radioactive. Figure 14 shows effluent quality through SBA in Cl– form after the bed has equilibrated (read that as totally exhausted) but continues to pull uranium. Ironically, even though highly selective, the uranyl ion can be regenerated off the SBA resin with conventional salt levels and reused. The regeneration cycle is usually calculated so the waste stream can be blended off with other waste streams to avoid being over the limit on radioactivity. Often this is in the 200,000-gal./cu. ft. range (~30,000 BVs). Before attempting to set up such a system, one should check with their resin supplier to determine the safe run limits without having the resin become a radioactive disposal issue. Under this limit, the resin can be safely disposed of into ordinary landfills.
Our final example of ion exchange versatility is the remineralizer. Figure 15 demonstrates what would happen if you had a stream where you wished to remove an ion with lesser selectivity than a partner ion with higher selectivity. In this case, we wish to remove Mg but not Ca. Glancing back at our selectivity listing, we see that Ca >>> Mg. If we use a softener to remove Mg, Ca comes along for the ride. But, we can put the Ca back in by simply passing the softened water through a bed of SAC that has been fully regenerated with a soluble calcium salt (i.e., CaCl2) to remove Na from the softened water and convert the soft water back to an all-Ca hardness stream. This can then be blended with the softened stream and a blended water containing no magnesium can be made. Clever, huh?
Conclusion
Ion exchange has been a proven method of separating minerals from water for over 100 years. In all likelihood, it will continue to be a preferred method for residential and industrial water treatment. With a little creativity and the right water, all sorts of things are possible.
References
1. Dow Chemical www.dowwaterandprocess.com/periodic_table
2. Michaud, C.F., Multi-Contaminant Removal in Municipal Systems. WC&P, May 2010.
3. Michaud, C.F., IEx-treme Softening. WC&P, June 2010.
4. Michaud, C.F., Achieving High-Purity DI Water Without the Use of Mixed Beds. WC&P, May 1997.





