by C.F. ‘Chubb’ Michaud
Hardness defined
The perception of soft water by commercial/industrial users goes well beyond simply preventing scale. Water that contains divalent calcium and magnesium ions is prone to leaving a hard residue behind upon heating or evaporation. We refer to such water as hard water. Hardness can be either temporary, which is associated with the weak-acid bicarbonate ion or permanent, which is associated with strong-acid anions, such as sulfate or chloride. Temporary hardness is so known because it can easily be reduced by simply heating the water and converting soluble bicarbonate salt to insoluble carbonate salt:
![]()
This explains why scale forms in boilers and dishwashers and why boiler water will often employ a de-alkalizer up front, along with a softener, to reduce both scaling and the evolution of an acidic steam (containing CO2) that can corrode pipes and downstream equipment. Softening alone does not reduce the corrosivity of steam produced from alkaline water.
Anion ‘softening’—effective scale control
The big issue is, should an operator encounter feedwater where the alkalinity exceeds that of the hardness and corrosivity, it is possible to condition the water with the use of a salt-regenerated anion only. This converts all of the anion to the chloride specie, and boiling the water will produce neither scale nor CO2 gas.
Scale formation
The bicarbonate salts of calcium and magnesium are soluble but do not exist as dry salts. Upon evaporation of water, even at ambient temperatures, bicarbonate converts to the insoluble carbonate. Hard water, simply left to dry (such as lawn sprinklers spraying onto a car) will form a hard and difficult-to-remove scale, which is why car washes hand-wipe water off metal and glass surfaces to prevent unsightly spotting. In addition, water evaporating from the surface of a swimming pool forms hard scale at the water line.
Permanent hardness reacts differently. It is not as prone to forming scale with heat, although gypsum scale (calcium sulfate) can form in high sulfate waters. Gypsum can also form when the high calcium sulfate solution is concentrated, such as at the surface of an RO membrane. Calcium chloride (CaCl2) salt is more water-soluble than is common salt, sodium chloride. CaCl2 does not precipitate under any condition but does not stick around long as a crystal because it is a deliquescent salt; that is, it will absorb enough moisture from the atmosphere to dissolve itself into a liquid. For this reason, it is often the salt of choice for sprinkling onto unpaved roads to hold down the dust. Water sources containing pure, permanent hardness would be very rare and would absorb enough CO2 from the air to convert to temporary hardness on exposure to the atmosphere. If total alkalinity exceeds total hardness, all temporary hardness results. If hardness exceeds alkalinity, both temporary and permanent hardness are present.
Both temporary and permanent hardness have adverse reactions with soap that form an insoluble soap curd, which we recognize as a bathtub ring. Soap is basically a sodium salt of an organic complex and is water-soluble. Divalent hardness salts are not very soluble. Soap acts as an ion exchanger and picks up the more highly selective divalent ion. Any time hard water and soap get together, enough soap to precipitate all of the hardness needs to be supplied first, and then enough to do the detergent job of cleaning. Precipitated soap and scum tend to be left behind, creating scratchy, rough fabrics and highly alkaline bed sheets, which can lead to skin irritation. In the shower, soap and shampoo seem to rinse off more easily with hard water. But does it really rinse off? No! It stays on the skin as an alkaline residue and can lead to irritation, skin rash and brittle hair.
Additionally, many cleaning products use alkaline boosters to raise the pH of the cleaning bath because oils and dirt are more easily rinsed off in higher pH water. The higher pH, however, also converts bicarbonate salt to a carbonate salt, which then precipitates. At even higher pH, all forms of hardness precipitate. This is why you cannot use hard water to regenerate the anion in a DI system using sodium hydroxide.
Personal use or industrial process water users are well aware of the difficulties encountered in using hard water. These include extra expense in equipment replacement due to scaling and extra labor required for removing deposits. Also, there are issues with restricted water flow from pipe scale and extra energy requirements in heaters and boilers. It is estimated that hard water at home adds 40 hours a year in extra labor.
One can summarize the advantages of using soft water by simply listing the disadvantages of using hard water:
- Hard water forms precipitate as carbonate scale when heated.
- Hardness forms precipitate with elevated pH.
- Hardness forms precipitate upon concentration.
- Hard scale forms on simple evaporation of hard water.
- Hard water reacts with soap to form insoluble soap curd.
Soft water defined
Softened water does none of these. By definition (by WQA Soft Water Task Force), soft water is that which contains less than one grain per gallon (gpg) of hardness ions (< 17.1 ppm as CaCO3). Any water conditioning device that is effective in reducing the water hardness to less than one gpg is, therefore, a softener. Any water conditioning device that does not reduce the hardness of the feed water to < 1.0 gpg cannot be called a softener, and the water it produces cannot be called soft water. A device that reduces the feed water hardness, but not to a level below one gpg is not a softener. There is no such thing as partially softened water, other than that obtained by blending soft water with hard water.
Alternative technologies needed
gy and use salt (NaCl) as the regeneration chemical. Great strides have been made to create a more efficient conventional softener (use less salt per gallon of water processed) but none have arrived at the perfect utility value of 6,000 grains recovered capacity per pound of salt (NaCl). There are those that use potassium chloride (KCl) in place of NaCl and call it saltless; however, KCl is a salt. It’s just not common or table salt (the name given specifically to NaCl a long, long, long time ago). Are there really devices that can be called saltless water softeners? Yes, there are. We know them as distillers (evaporative), reverse osmosis and nanofiltration systems (membrane), de-ionizers (ion exchange), capacitive deionization (EDI) systems and, of course, portable exchange softener tanks (at the point of use).

There is a tremendous push today to achieve softened water without the use of salt. The primary driver for this is the need to maintain low TDS in wastewater so it can be recycled or reprocessed for groundwater injection (without the need to reduce its TDS with additional expensive equipment). This issue has brought about several laws restricting the use of salt-regenerated water softeners in the state of California, and there are active programs of study that may do the same in Arizona and Nevada. Is there an alternative to the salt-regenerated water softener?
Scale prevention
Perhaps the biggest detriment to using hard water is the formation of scale. There are several technologies in use today that work toward scale reduction. The first and most widely used technology is the removal of scale-forming ions by using a conventional salt-regenerated ion exchange softener. There are times when there is a need to remove most everything in the water and go toward demineralization (which also softens the water). Next, there are means of treating water with chemical addition to either prevent the formation of scale, or at least make scale in a softer and more easily removed form. Scale preventers work by sequestering hardness with chemical additives (chelating agents) that effectively tie up hardness to make it non-reactive. EDTA (ethylenediamenetetraacetic acid) is such an additive. Other additives (such as low levels of polyphosphate) induce a soft scale by interfering with the crystal lattice of hard-scale formation.
Finally, there is a host of physical water treating (PWT) devices that neither remove scale-forming ions nor add chemicals. Some are scale-inducing devices, which form microscopic crystals of hard scale in the water phase that prevent scale from building on surfaces and interfering with flow and heat transfer; and hard-scale-preventing devices that convert the scale specie from the hard calcite to aragonite or some other form of soft scale that is more easily removed. All of the above mentioned technologies have demonstrated, to some degree, that they are capable of reducing problems associated with the accumulation of hard scale. In our list of problems with hard water mentioned earlier, these technologies address scale prevention.
Options for commercial and industrial water treatment
If the primary issue at hand is the prevention of boiler scale with subsequent loss of heat transfer efficiency and the ensuing maintenance and labor costs associated with extensive cleaning, most of the above technologies will find some level of acceptance. Some are more readily accepted than others. The formation of soft scale by some PWTs, for instance, still requires periodic shutdowns and cleaning. The same is true of some chemical treatments (crystal dispersion). Options 1 and 2, both of which actually remove the hardness ions, create water that is non-scaling.
Deriving soft-water benefits
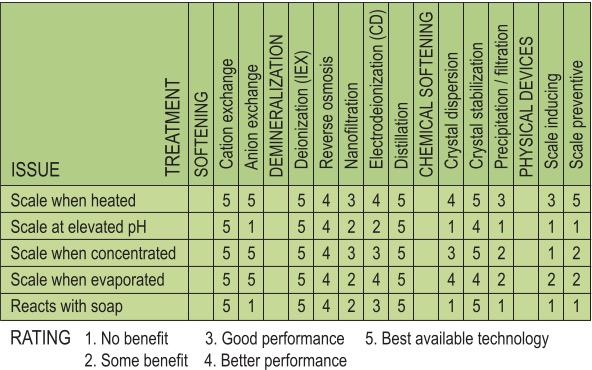
If all of the benefits of soft water need to be achieved with your water treatment device, choices are much slimmer. Divalent ion reactivity will have to be removed from the water by one of the removal methods listed in Table 1. This can only be done by removing divalent (hardness) ions (by softening or demineralizing), or by using a chemical chelating agent such as EDTA to prevent reaction. Table 2 summarizes the benefits of each.
Discussion of Table 2
In order for any of the adverse reactions to occur, there must be hardness ions present in sufficient quantity in order to be noticeable. Cation exchange softening, deionization and distillation are the only processes that essentially remove divalent ions to near zero. Anion exchange removes counter ions (bicarbonates and sulfates) so hardness will not form scale, but leaves hardness ions, which are free to react with soap or precipitate at elevated pH (as hydroxides).
Reverse osmosis (at low pressure) is not 100-percent effective at rejecting calcium and magnesium; in a typical 300-ppm hard feedwater, it will leave from four to eight ppm of hardness—enough to cause noticeable scaling. Nanofiltration has even lower rejection rates for hardness. Capacitive DI (those newer devices that remove hardness through electrical attraction with DC current) removes sufficient hardness to reduce scale by about 95 percent1 but leave sufficient hardness so that the water will still react with soap and cause severe clouding at high pH.
Chemical chelation essentially locks up divalent ions so they cannot react; therefore, they do not form scale when heated or evaporated, and the water will not react with soap. This type of additive (EDTA) is often incorporated into cleaning compounds for the express purpose of allowing soap to do its job in hard water. To a lesser extent, those chemical additives that allow scale to form a precipitate, but interfere with scale growth by dispersing the micro-particles, allow a high amount of reactive hardness to remain in solution. The combination of precipitated scale and new, hard scale leaves a deposit that is considerably softer and more easily removed. There is still sufficient reactive hardness in solution to react with soap and cause clouding at elevated pH.
Lime softening usually requires ion exchange polishing for most applications but does reduce hardness down to three or four grains. Hot lime will reduce hardness to below one gpg but is not suitable for anything other than large industrial or municipal applications, due to the chemical complexity and size of the equipment.
Physical water treatment falls into two general categories: scale-inducing technology, which works by encouraging formation of scale before a water heater in order to prevent the formation of scale in the water heater. This group includes template assisted crystallization (TAC), which catalyzes the formation of sub-micron crystals that remain in the water as colloidially suspended particles (which do not stick to heated surfaces) and electrically induced precipitation (which forms a soft scale on an electrode that must be periodically cleaned). The second group is the scale prevention technology, which purports to modify water in order to prevent scale formation. This group includes capacitive deionization (which actually reduces the water’s ionic load by attracting soluble ions to selective electrodes), and electromagnetic devices that claim to form a soft precipitate consisting of amorphous aragonite and vaterite crystals, instead of the more tenacious hard-scale form known as calcite(2).
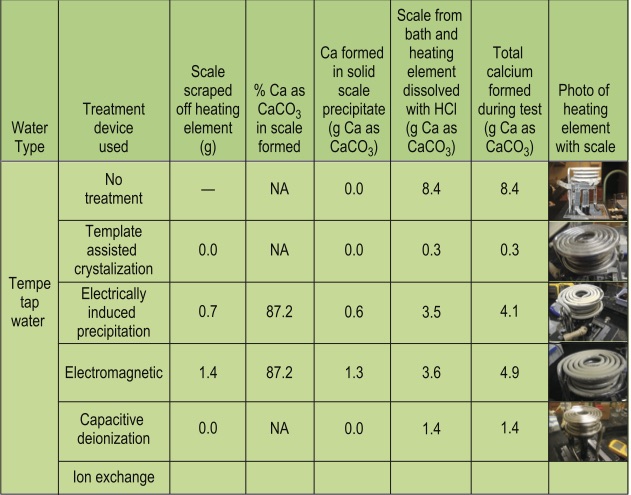
At the November 2010 meeting of the Arizona WQA, Dr. Peter Fox, Professor of Environmental Engineering, Arizona State University, Tempe, presented a paper with interim results on a study he is conducting on alternative water treatment devices.2 Specifically, his study is an evaluation of alternatives to salt-regenerated water softeners, with an interest in domestic applications. The devices selected were based on a variety of different technologies with reports of a good success history. Dr. Fox’s study centered on the German Association of Gas and Water (DVGW, using test protocol W512) methodology for evaluating alternative devices and therefore, focused on scale prevention only. This test processes treated and untreated hard water through an electric water heater for 20 days, then quantifies the scale formed in the heater. Scale that forms is measured and compared to untreated water and reported as a percentage reduction. In order for a device to ’pass‘ and be certified by DVGW, it must reduce the scale formation by 80 percent or more. The devices tested included:
Template Assisted Crystallization (TAC)
TAC employs a special surface-treated resin bead to act as a catalytic nucleation site for the conversion and growth of hardness crystals, which are subsequently released and remain in solution without forming scale (DVGW certified).
Electrically induced precipitation
This process uses direct current to form soft precipitate onto an electrode, and subsequently requires a backwash to clean the electrode (DVGW certified).
Capacitive deionization
An old technology with a new twist, using DC current to separate ions in water into positive and negative species and retains them on an electrode. Reduces TDS and hardness of the water. Regenerated by reversal of current, with occasional chemical cleaning. Meets DVGW-certification criteria for scale prevention.
Electromagnetic technology
These devices consist of a wire wrapped around the water pipe that transmits electronic frequencies (magnetic field) and claims to precipitate hardness into aragonite crystal, forming a soft scale as opposed to calcite. The exact mechanism is not clear but it is theorized that dissolved oxygen, silica and carbonate ion may play a role.
Summary of interim test results
Table 3 (from Dr. Fox’s presentation at AWQA) lists the devices, where scale was found (on heating elements or in heater tanks) and the total scale formed, along with a picture of the heating element after the test. The scale that could be measured appeared as 87.2-percent calcium. To calculate effectiveness of the treatment, results are compared to non-treated water (Tempe, AZ tap water from the Salt River, with average hardness of 180 ppm as CaCO3). The heaters used in the study had a watt density of 25 W/cm2, which is closer to the actual power of an electric water heater and differs from the actual DVGW protocol.
Bottom line
Table 4 shows calculation of the percent of scale reduction tested for the various technologies. It should be pointed out that only one aspect of scale prevention was tested: the effects on the scaling tendency of hard water subjected to heating. With the exception of the untreated feedwater, the scale formed was soft and easily removed. Scale accumulation, however, would be problematic.
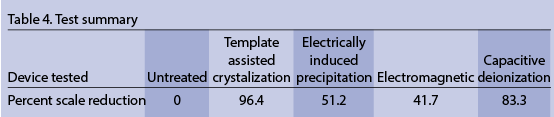
As summarized in Table 2, there are other benefits to removing hardness from water through actual softening. Table 2 also estimates the results of testing, if it was carried out on other aspects of hard-water reactions. Bottom line: alternative treatment devices are not water softeners. For many industrial applications requiring scale prevention only, they offer an alternative technology to softened water but are not alternatives to softeners.
Continued study
Dr. Fox’s study, funded by the Water Research Foundation, is not yet complete. There are additional water sources and devices to be tested, including the salt-regenerated ion exchange softener.
References
1. Nanofiltration, Dow Chemical Water & Process Solutions, www.dowwaterandprocess.com/products/ronf.htm
2. Fox, Peter, Evaluation of Alternatives to Domestic Ion Exchange Water Softeners, WRF-08-06. AWQA luncheon presentation, November 19, 2010, referenced with permission.





